Lab IT Compliance and Validation of Computerized Laboratory Systems
We support you on the way
to the digital transformation of your laboratory!

Digitization in the laboratory
This is the topic we deal with in our new whitepaper “Digitization of laboratories in the life sciences – still plenty of room for improvement”. According to a study by Fraunhofer IPA, there is still a lot of potential for optimization in the digitization of laboratory processes. We often still encounter many analog processes and activities in everyday laboratory work that could be supported and accelerated with the help of IT tools and systems or even technologies such as augmented reality. In addition, isolated solutions with laboratory devices and systems from a wide range of manufacturers that do not communicate with each other, or only communicate with each other to a limited extent, must be eliminated.
The tasks and issues involved in increasing the degree of digitization, automation and networking in your laboratory are complex and multifaceted.
Of course, lab executives and employees want to continue to drive digitization. “However, reality stands in the way of this desire,” says study author Milena Frahm of the Laboratory Automation and Bioproduction Technology department at Fraunhofer IPA. “In many laboratories, digitization is progressing so slowly mainly because of a lack of time, money and interfaces.” Respondents saw another major obstacle in gaining the acceptance of all employees for digital solutions. In addition, many laboratory employees and managers said they knew too little about the technical possibilities of new technologies to be able to identify meaningful IT application areas for laboratories.
This is where IT specialists – like those at DHC – are needed, who not only have the necessary IT know-how on systems and equipment, but also bring in-depth knowledge of laboratory processes, IT compliance issues, validation and qualification, and the specific industry requirements. After all, despite the digitization initiative, your lab team should still have priority time for its value-added activities.
* The study “Digitalization status and digital reality in life science laboratories” is available for free download at:
https://www.ipa.fraunhofer.de/de/Publikationen/studien/digitalisierungsstand-von-life-science-laboren.html
What needs to be done and how can DHC support here?
How can the digitization of laboratories be driven forward?
Digitization in the lab can be driven in a number of ways.
Here are some examples that can help:
Introducing Laboratory Information Management Systems (LIMS)
LIMS are purpose-built software solutions that automate the management of laboratory processes, samples, analyses and data. By implementing a LIMS, laboratory results can be efficiently recorded, managed and shared.
Automation of laboratory processes
Routine laboratory tasks can be automated through the use of robots and automated systems. This increases efficiency, reduces errors, and allows experiments and analyses to be performed more quickly.
Use of data analysis and artificial intelligence
Digitization enables the collection of large amounts of data in the laboratory. By using data analysis methods and artificial intelligence, patterns and relationships in the data can be identified to generate insights and predictions. This can increase the effectiveness of research and development in the lab.
Implementation of electronic laboratory notebooks (ELNs)
ELNs replace traditional paper lab notebooks with electronic records. These allow for better organization, searching, and sharing of laboratory information. ELNs also provide the ability to link data directly to other systems, such as LIMS or data analysis tools.
Networking and integration of systems
Digitization in the laboratory requires the integration of various systems to enable seamless data flow and communication. This includes the integration of measurement devices, analysis software, LIMS, ELNs and other relevant systems.
Staff training and education
To drive digitization in the laboratory, it is important that laboratory staff have the necessary digital skills. Training and education should be provided to ensure that staff are familiar with digital tools and technologies and can use them effectively.
These approaches can help drive digitization in the laboratory and increase the efficiency, accuracy, and productivity of laboratory work. It is important to consider the specific needs and goals of the lab and choose appropriate digital solutions to achieve the best possible results.”
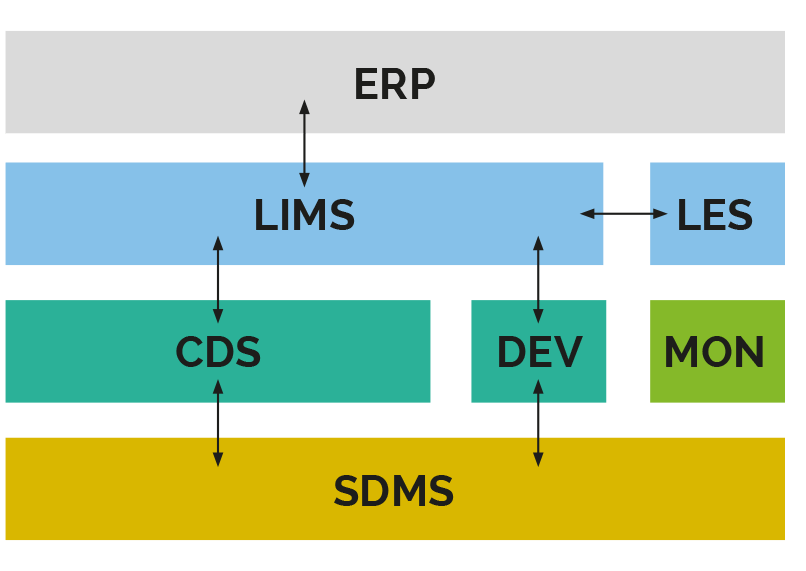
SYSTEM LANDSCAPE IN THE TESTING LABORATORY
Support of resource planning and various workflows of the entire company (e.g. SAP S/4HANA).
Support of workflows and transparent tracking of samples over the entire processing cycle in sample-oriented laboratories with defined workflows (e.g. LabWare LIMS).
Specialized variant of the ELN to support and document laboratory activities according to work instructions in sample-oriented laboratories with defined workflows (e.g. STARLIMS LES).
Control of chromatographs and acquisition and evaluation of chromatography data (e.g. Waters Empower).
Support for sample preparation and sample analysis directly or via intermediate layer (e.g. scales, pH meter, IR spectrometer).
Continuous monitoring of climatic ambient conditions such as humidity and temperature (e.g. testo Saveris 2).
Archiving of data from other laboratory systems, enabling centralized data storage and support for data analysis (e.g. Thermo Scientific SampleManager SDMS).
Thursday 23 November 2023, 10:00 – 10:45 a.m.
WHY CHOOSE DHC?
Make use of our experience and industry expertise as validation and data integrity specialists:
- Over 25 years of expertise in computer system validation
- A wealth of expertise in regulatory requirements
- Broad-based experience with a very wide range of IT systems
- Integrative approach adopted for existing QM systems
- High level of knowledge transfer
Managing Director & Industry Unit Manager
Life Sciences, Chemie and Medical Engineering
phone: +49 681 93 666 0
email: matthias.bothe@dhc-gmbh.com

CEO and Director Sales & Marketing, DHC Switzerland
phone: +41 44 500 888 7
email: carsten.buri@dhc-ag.ch

Learn more about our consulting services for GxP Compliance!
 Deutsch
Deutsch Schwyzerdütsch
Schwyzerdütsch English
English








